Abstract
Background: Randomized trials have shown a beneficial effect of anticoagulation with warfarin to prevent stroke in atrial fibrillation. It is not known whether the same effect will be obtained in actual practice. The authors conducted a prospective observational study to evaluate the effect of preventive anticoagulation in patients with atrial fibrillation in 2 practice settings in Montreal.
Methods: Of the 1725 outpatients screened between October 1990 and September 1993 at a community hospital and a university-affiliated hospital, 221 with documented atrial fibrillation were enrolled and followed up for a mean of 27 months. Most (75%) of the patients excluded did not meet the inclusion criteria (because of, for example, an artificial heart valve, mitral stenosis, cardiac transplantation or transient atrial fibrillation); the remainder had not completed enrolment before the end of the study. Following the baseline visit, patients were interviewed by telephone every 6 months, and reported events were confirmed through review of the patients' charts. Hazards for stroke and for stroke and transient ischemic attack (TIA) combined were calculated for each of 4 treatment groups: ASA, warfarin, blended treatment and no treatment, based on the type of anticoagulation therapy patients received during the entire observation period. The blended-treatment group consisted of patients who started on one active therapy and switched to the other or who switched treatments more than once. Corresponding rate ratios (RRs) and 95% confidence intervals (CIs) were calculated with reference to the no-treatment group. Cox proportional hazards analysis was used to adjust for differences in patient characteristics. The rates of bleeding episodes were also analysed.
Results: On average, the study patients were older (71.6 [standard deviation 9.3] years) and had a higher prevalence of underlying heart disease (52.0%) than those in the randomized trials. Nineteen patients had a first stroke: 4 in the ASA group, 4 in the warfarin group, 4 in the blended-treatment group and 7 in the no-treatment group, for rates of 5.2, 1.8, 5.3 and 5.9 per 100 person-years, respectively. Only warfarin was associated with a significantly lower risk of stroke compared with no anticoagulant therapy (RR 0.31, 95% CI 0.09-1.00). A similar protective effect of warfarin was found for stroke and TIA combined (2.3 v. 6.7 per 100 person-years; RR 0.34, 95% CI 0.12-0.99); the effect of ASA and blended treatment was not significantly different from no treatment. The rate per 100 person-years of any bleeding was not significantly higher for any treatment group (ASA 2.5, warfarin 3.4 and blended treatment 3.5) compared with the no-treatment group (1.9). Patients receiving warfarin had a significantly greater risk of any bleeding event than patients not receiving anticoagulant therapy (RR 1.79, 95% CI 1.07-3.00).
Interpretation: The relative effect of anticoagulant therapy with warfarin in preventing stroke in these practice settings was equivalent to that in the randomized trials, although these patients were older and sicker. This preventive treatment is likely to confer additional benefit as it is more widely prescribed.
People with atrial fibrillation are at increased risk for stroke, presumably from cardiac emboli. [1, 2] Randomized trials have demonstrated an effect of warfarin in preventing such strokes. [3–8] Of the 3 trials that evaluated ASA [4, 5, 9] one showed it to be of benefit as well, although to a much lesser degree. [4] In an overview of these trial results, we hypothesized that warfarin would be similarly effective in actual clinical practice, [10] although this has not been studied. Despite the encouraging trial results, subsequent studies have shown that warfarin is being prescribed for only about two-thirds of patients with atrial fibrillation. [11–15] Among the possible reasons for this underuse is the problem of translating randomized trial results to the actual practice setting.
We conducted an observational study in the years following the publication of the warfarin clinical trial results to evaluate whether a similar protective benefit would be obtained in actual practice. To evaluate the effect of preventive anticoagulant therapy, we studied 2 practice settings in a major Canadian city: a community hospital with a local clientele and a university-affiliated hospital with a referral clientele.
Methods
All patients who underwent electrocardiography (ECG) during any outpatient visit were screened between October 1990 and September 1993 at the Royal Victoria Hospital, a 650-bed teaching hospital, and the Reddy Memorial Hospital, a 300-bed community hospital, both in Montreal. Eligible patients were required to have atrial fibrillation documented on at least 2 ECGs obtained more than 30 days apart. For all such patients, further information was sought from the referring source and from the patient's hospital chart. Patients were excluded if they had mitral stenosis, an artificial heart valve or cardiac transplantation. For those not excluded at this first stage, the patient's physician was contacted for further assessment of eligibility. Patients could be excluded at this second stage because of refusal to participate, severe illness (e.g., disabling mental illness or end-stage cancer), atrial flutter or resolved or transient atrial fibrillation, severe language barrier, inability to be followed up (e.g., imminent plans to relocate or residence too far from hospital) or death in the interim.
At the enrolment visit we obtained baseline demographic data and information about concomitant conditions and risk factors. These included information on age, sex, weight, height, employment status and the presence of any physician-diagnosed hypertension, hypercholesterolemia, congestive heart failure, diabetes mellitus, angina, myocardial infarction or previous stroke. Current anticoagulant therapy and treatment for diabetes, angina, hypertension or atrial fibrillation were documented. Echocardiogram data were also obtained.
Follow-up ECG and telephone interviews were conducted every 6 months. We obtained information on risk factors for stroke and noted any change in anticoagulant medication. The patient was asked about the occurrence in the interim of physician-diagnosed stroke, other emboli, transient ischemic attack (TIA), intracranial hemorrhage, hemorrhage or any hospital admission. When outcome events were reported, we obtained confirmation and documentation from the patient's chart. Stroke and intracranial hemorrhage were documented by CT scanning in many cases. We classified all hemorrhages on an ordinal severity scale based on the amount of concern it caused on the part of the patient or the action that was taken. Bleeding was classified as major if it was fatal (death due to the hemorrhage), if surgery or packing was required or if a transfusion was required.
We included in the analyses the data for all enrolled patients with follow-up information. We compared treatment groups with respect to baseline demographic characteristics, presence of relevant concomitant conditions and presence of risk factors for stroke using chi squared tests for categoric variables and analysis of variance for continuous variables.
For each patient, we calculated the number of person-years of observation as the length of time between the enrolment interview and the last follow-up interview. For the analysis of first occurrence of outcome events, person-years for an individual patient were censored at the time of the first occurrence or the end of follow-up (June 1994), whichever came first. This was calculated separately for each type of outcome event.
Patients who received either one type of treatment or no treatment for the entire observation time were assigned to the ASA, warfarin or no-treatment group accordingly. The remaining patients who switched treatments were assigned to the blended-treatment group. For patients who switched only once from no treatment to ASA or warfarin, or vice versa, the time at risk was allocated to the associated treatment status.
We calculated hazards for each outcome event as the number of events divided by the total number of person-years accumulated for each treatment group. To assess the association between outcome and treatment for first events, we calculated rate ratios (RRs) (with 95% confidence intervals [CIs]) for stroke, stroke and TIA combined, all hemorrhages and major hemorrhages, using the no-treatment group as the reference group. Since patients were followed for different lengths of time, we used Kaplan-Meier analyses to estimate the cumulative rate of remaining event-free, with patients being censored at the time of the first outcome event or the end of follow-up, whichever came first. We carried out a Cox proportional hazards analyses to account for possible confounding.
The study was approved by the ethics committees of the 2 hospitals. We obtained permission to contact an eligible patient from the physician who had ordered the initial ECG or from the primary care physician, as determined from the patient record. Patients gave informed consent at the enrolment visit. Study records and findings were kept separate from the hospital records but were forwarded to the patient's physician after the patient's approval was obtained.
Results
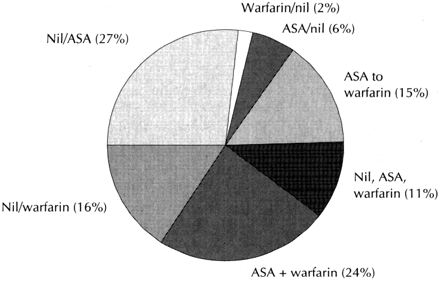
Of the 1725 patients who had atrial fibrillation on screening ECG, 1504 (87%) were not enrolled: 375 (22%) were ineligible at the first stage (because of mitral stenosis, artificial heart valve or cardiac transplantation) and 756 (44%) at the second stage (for reasons such as transient atrial fibrillation, death in the interim or language barrier). An additional 373 patients (22%) had not completed the second stage of the enrolment procedure by the time the study ended. The remaining 221 patients were enrolled and followed for an average of 26.8 (range 4.0-48.8) months, providing a total of 493.7 person-years of follow-up as of June 1994. Of the 221 patients, 31 (14%) received ASA as treatment for their atrial fibrillation during the entire follow-up period, 87 (39%) received warfarin, 65 (29%) received some combination of ASA, warfarin and no treatment (Figure 1), and 38 (17%) did not receive anticoagulant therapy.
Figure 1. Distribution of treatment among 65 patients with atrial fibrillation who switched treatment groups during the observation time (blended-treatment group).
The demographic characteristics and the presence of relevant concomitant illnesses and risk factors for stroke are shown in Table 1 for the 4 treatment groups. The mean age for all the patients was 71.6 years, and the mean length of time since the diagnosis of atrial fibrillation was 7.7 years. Over half of the patients were men. There were no significant differences between the groups in age, sex, duration of atrial fibrillation, or presence of angina, myocardial infarction, congestive heart failure, hypertension or hypercholesterolemia. A higher proportion of patients in the warfarin group and the blended-treatment group than in the ASA group and the no-treatment group had diabetes or a history of a previous stroke (p < 0.001).
Table 1. Characteristics of patients with atrial fibrillation from 2 practice settings, by treatment
Nineteen patients had strokes during the study period; 1 patient in the no-treatment group had 2 strokes (Table 2). Six patients had at least 1 TIA during the study period; 1 patient in the blended-treatment group also had a stroke. Patients receiving warfarin had a significantly lower risk of stroke than those who received no treatment (RR 0.31, 95% CI 0.09-1.00). A similar protective effect of warfarin was found with stroke and TIA combined (RR 0.34, 95% CI 0.12-0.99). When the analysis was limited to patients without a previous stroke, the same effect was found (RR 0.22, 95% CI 0.05-0.96). The effect of ASA (RR 1.04, 95% CI 0.17-6.15) and blended treatment (RR 1.06, 95% CI 0.23-4.89) was not significantly different from no treatment.
Table 2. Rates of first occurrence of stroke, stroke and transient ischemic attack (TIA) combined, and any bleeding episode for the 4 treatment groups
Kaplan-Meier analyses showed a significant difference in the median time to stroke between the warfarin group and the no-treatment group (24.9 months v. 18.4 months) (p = 0.01). This difference remained significant (p = 0.012) when selected demographic and risk factor variables were included in the Cox proportional hazards model. No significant difference was found between the ASA and no-treatment groups.
Five patients, 4 in the warfarin group and 1 in the no-treatment group, had major hemorrhages during the study. The patient in the latter group experienced 2 major hemorrhages: the first necessitated a transfusion, and the second was fatal. One patient in the warfarin group died from the hemorrhage, 2 required surgery or packing, and 1 required a transfusion. The rate of major bleeding was not significantly greater for any group compared with the no-treatment group, although the lack of significance for the warfarin group (RR 2.4, 95% CI 0.3-22.9) was likely due to the small sample. Patients receiving warfarin had a significantly greater risk of experiencing any bleeding event than those in the no-treatment group (RR 1.79, 95% CI 1.07-3.00) (Table 2).
Interpretation
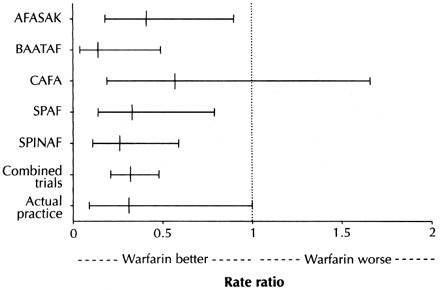
The essential finding of our study is that the protective effect of warfarin against stroke in patients with atrial fibrillation demonstrated in randomized trials [3–8] translates well to the actual practice settings of this study (Figure 2).
Figure 2. Efficacy of warfarin in preventing stroke in patients with atrial fibrillation in clinical trials versus actual practice. AFASAK = Atrial Fibrillation, Aspirin, Anticoagulation Study; [5] BAATAF = Boston Area Anticoagulation Trial for Atrial Fibrillation Study; [6] CAFA = Canadian Atrial Fibrillation Anticoagulation Study; [7] SPAF = Stroke Prevention in Atrial Fibrillation Study; [4] SPINAF = Veterans Affairs Stroke Prevention in Nonrheumatic Atrial Fibrillation Study; [8] combined trials: AFASAK + BAATAF + CAFA + SPAF + SPINAF; actual practice: present study.
The patients in our study were much more representative of the atrial fibrillation population than were the patients enrolled in the randomized trials. The main differences are that the patients in our study were, on average, 2 years older and had a higher prevalence of serious concomitant conditions (myocardial infarction 22.2% v. 12%, and congestive heart failure 30.8% v. 19%). The choice of preventive therapy was left up to the individual practitioner at a time when the trial results were generally known. Thus, our study ought to be a good assessment of the potential benefit of warfarin anticoagulant therapy in actual practice settings.
In our study, warfarin increased the number of major bleeding episodes by 11 per 1000 person-years compared with no treatment; the increase in the trial overview [3] was 3 per 1000 person-years compared with placebo. However, major bleeding episodes were few in our study, which makes the estimate imprecise. It is thus not possible to conclude definitively that warfarin would be less safe in actual practice than in the closer surveillance conditions of the randomized trials. An analysis of the bleeding experience from 2 trials shows that the risk of bleeding depends on the degree of anticoagulation, [16, 17] a feature that our study was not designed to measure.
There was a small but statistically insignificant protective effect of ASA. An overview of 3 trials that compared ASA with placebo found only a small effect of borderline statistical significance. [18] Although our failure to find a statistically significant effect may be the result of the size of the study, the RRs are quite different from those found with warfarin. In fact, the RR for ASA in our study, 0.78 (95% CI 0.24-2.50), is remarkably similar to that of the overview (0.79, 95% CI 0.6-1.0). [18]
The strengths of our study are its prospective design, regular surveillance for the presence of atrial fibrillation and treatment, and documented outcome events. It was conducted in 2 practice settings free of the selectivity and constraints of a randomized trial. The main limitation is its size. Thus, further evaluation of bleeding rates is required.
Given the estimates of treatment effect in the randomized trials together with the effect observed in actual practice, warfarin anticoagulant therapy should be offered with confidence to patients with atrial fibrillation. A greater proportion of patients will likely benefit than the proportion currently estimated to be receiving warfarin treatment. [11–15]
This work was supported by grants from the Quebec Heart and Stroke Foundation, the Bank of Montreal, the Research Fund of the Division of General Internal Medicine, Royal Victoria Hospital, Montreal.
Competing interests: Dr. Caro has received grants from pharmaceutical companies for conducting research in this area. The other authors declared no competing interests.
This article has been peer reviewed.
Reprint requests to: Dr. J. Jaime Caro, 336 Baker Ave., Concord MA 01742, USA; fax 978 371-2445; [email protected]